Author(s): Susmita1, Ravi Kant, Prakash Tendulkar, Parshika , Monika Pathania
Severe acute respiratory syndrome coronavirus 2 (SARS CoV-2) has been a global pandemic that was first reported in December 2019. Since then, it has been a subject of study for all researchers about its effects in various organ systems of the human body. SARS CoV-2 infection has been associated with Myocarditis in many cases. However, there are only a few instances where COVID 19 was associated with Pericarditis, which is too constrictive. In the following case, we shall discuss a previously healthy 23 years old young man who developed Constrictive Pericarditis as sequelae of COVID 19 Pneumonia.
The spectrum of SARS-CoV-2 infection ranges from asymptomatic to critical illness with respiratory failure and multiorgan failure. Cardiovascular involvement can be in the form of an asymptomatic increase in myocardial injury markers, contractility disorders, arrhythmias, pericardial disease, vascular insufficiency and sudden cardiac arrest [1].
Although the pathophysiology of Pericarditis in COVID-19 is unknown, it is hypothesized that it occurs secondary to the systemic inflammatory response and cytotoxic and immunemediated effects related to SARS-CoV-2 [2]. Proinflammatory cytokines (IL-6, TGF-β) and vascular endothelial growth factor (VEGF) released via the associated angiotensin II pathway could promote inflammation and fibrosis of the pericardium in COVID-19 infection [3,4].
We report the case of a young male patient with SARS-CoV-2 infection who developed constrictive Pericarditis in the course of the disease. This case highlights the importance of high clinical suspicion of pericardial complications of COVID-19.
A 23 years old male with no known co-morbidities had a history of low grade, intermittent fever three months back, and was diagnosed with mild covid-19. He presented with a complaint of low-grade fever 20 days back for five days and generalized body swelling for the last 15 days. Fever was low grade, intermittent and resolved with over-the-counter antipyretics. Generalized body swelling was gradual, progressive. Initially, it was noticed on the abdomen and gradually progressed to the whole body and was associated with orthopnea and exertional dyspnea. There was no associated decreased urine output, cough, sputum and Malena. There was no history of tuberculosis (TB) or TB contact, malignancy, any other chronic disease in the past. There was no history of surgery in the past as well.
On examination, he had a generalized pitting type of oedema along with engorged neck veins. Kussmaul’s sign and Pulses paradoxus was also present. The abdomen was distended, and fluid thrill could be elicited; no organomegaly was appreciated. He also had bilaterally decreased air entry on respiratory examination and dull note on percussion, especially more in the right than the left.
For these complaints, he was started on anti-tubercular treatment (ATT) by an outside hospital based on clinical suspicion, and he took ATT for ten days. On further investigation, ECG showed low voltage complexes and generalized T wave flattening and inversion. (Figure-1) Chest x-ray was suggestive of bilateral pleural effusion right more than left. (Figure-2) HRCT thorax (3/6/21) showed ground-glass opacities in the lower lobe of the left lung and CORADS 4; b/l moderate pleural effusion; mild pericardial effusion. ATT was stopped, and the patient was managed symptomatically and in line with heart failure. On presentation, he had transaminitis, hyperbilirubinemia and Hyperuricemia. Hyperuricemia, which was attributed to ATT, and was stopped. Diagnostic Pleural fluid and ascitic fluid analysis were done and was found to be transudative fluid.
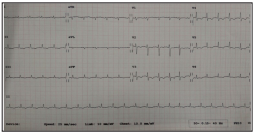
Figure 1: ECG suggestive of low voltage complexes
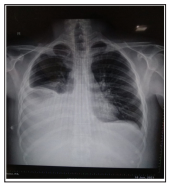
Figure 2: Chest x-ray s/o bilateral pleural effusion (Right>left)
Both fluids were culture negative and CBNAAT negative. Sputum induction was done and sent for AFB and CBNAAT, which came negative. TSH was 3.9 mIU/L, NT pro-BNP was 151.9 pg/ml, and Trop I was negative. ESR was also come normal (3 mm/hr). Screening ultrasound whole abdomen was done, and it showed hepatomegaly (16.6cm) with raised echotexture and moderate ascites. 2 D ECHO was done, which showed a slightly reduced left ventricular ejection fraction of 50-55 % with grade 2 left ventricle dysfunction and thickened pericardium. (Figure-3)
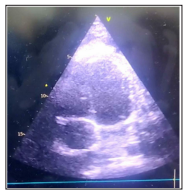
Figure 3: Transthoracic ECHO s/o cardiac wall thickening
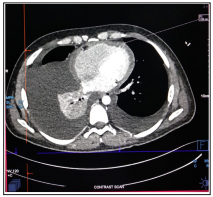
Figure 4: CECT thorax s/o cardiac wall thickening (1.03)
Other findings on echocardiography were respiration related ventricular septal shift with E-velocity variation of > 25% on the mitral valve and >35% on the tricuspid valve. Inferior vena cava was distended and non-collapsible. It was followed by CECT thorax (Figure-4), suggestive of b/l pleural effusion, cardiomegaly, moderate ascites with subcentrimatric mesenteric lymph nodes. The patient was diagnosed with chronic constrictive Pericarditis secondary to COVID pneumonia. ATT induced hepatitis; Hyperuricemia was resolved gradually after stopping ATT. The patient has been managed symptomatically with diuretics and other supportive treatments. He improved with the treatment and was discharged in hemodynamically stable condition and advised to follow up with a cardiology and cardiothoracic vascular surgeon (CTVS).
Investigations: (Table-1)
| 3/6/21 | 4/6/21 | 7/6/21 | 11/6/21 | 14/06/2021 | |
| Hb(gm/dl) | 14.45 | 12.6 | 13 | ||
| TLC (1000/ul) | 8.47 | 8.5 | 4.84 | ||
| DC (N/L) | 64/22 | 90/12 | 78.5/15 | ||
| Platelet(Thousand/ul) | 98 | 101 | 121 | ||
| T.B/D. B (mg/dl) | 3/1.51 | 2.85/1.9 | 2.61/1.12 | ||
| SGOT/SGPT (U/L) | 309/164 | 248/168 | 74/76 | ||
| ALP(IU/L) | 119 | 115 | 66 | ||
| T. Protein/alb (g/dl) | 6.36/2.85 | 5.9/3.34 | 6/2.9 | ||
| Na/K(mEq/l)/Ca(mg/ dl) | 133/5.5/8.57 | 127/3.74/8.43 | 129/3.77/8.6 | 132/4.14 | |
| B. urea/creat (mg/dl) | 27/0.82 | 24.8/0.76 | 16/0.62 | 18/0.76 | |
| Uric acid/ phosphorus (mg/dl) | 17/3.35 | 20.16/2.71 | 12.5/1.7 | 7.8/2.7 | |
| HIV/HCV/HBV | negative | ||||
| TSH (m IU/L) | 3.9 |
ASCITIC FLUID: TLC- 90 Cells, DLC- 75% Neutrophil, LDH 152, AMYLASE: 7, ADA: 4.85 U/L
PLEURAL FLUID: TLC- 140cells/ml, DLC- 60% Neutrophil, Protein- 0.6 g/dl, ADA: 10 U/L, CBNAAT: Negative
SPUTUM- CBNAAT: Negative, AFB: Negative
ESR: 03 mm/hr, NT pro BNP: 151.96 pg/ml
SARS CoV 2 infection is remaining a significant concern due to his high infectivity and mortality. Respiratory involvement leading to severe acute respiratory distress syndrome (ARDS) is the primary and leading cause of mortality due to Covid-19. The virus binds to angiotensin-converting enzyme 2 (ACE 2) receptors through viral coat protein SPIKE (S protein); it is followed by priming by the serine protease TMPRSS2 mediated virus uptake. ACE 2 receptor is expressed in all tissues but primarily in the lung, kidney, heart, vessels, gut and brain. Hence, Covid-19 can cause cardiovascular complications like Myocarditis, pericardial effusion and Pericarditis [5].
Constrictive Pericarditis (CP) can occur after any pericardial disease [6]. Constrictive pericarditis is characterized by the inflammatory and fibrotic thickening of the parietal and visceral pericardium, leading to impaired diastolic filling of the ventricles due to pericardial disease, with signs and symptoms of right heart failure with preserved right and left ventricular function in the absence of concomitant or previous myocardial disease or advanced forms. The most common causes of constrictive Pericarditis are bacterial and tuberculous Pericarditis, radiotherapy and cardiac surgery. Idiopathic and viral Pericarditis has a low incidence of constrictive Pericarditis [7].
In a case report published by Rodica Daiconu et al. in May 2021, a previously healthy 27-year-old woman developed a severe form of COVID-19 with massive pneumonia. Within 2-3 weeks, it was followed by acute Pericarditis in which constrictive physiology developed rapidly [8].
For diagnosing constrictive Pericarditis, imaging modalities include echocardiography, CT, MRI, and cardia catheterization studies. It is necessary to screen for the presence of any cardiac complications, pericardial effusion in patients with COVID -19 pneumonia. It is thus essential to study various courses and complications of this pandemic, including pericardial complications that can lead to high morbidity and mortality [9].
If early management of pericardial effusion and acute Pericarditis is done, it rarely develops into constrictive Pericarditis [10]. In chronic constrictive Pericarditis, pericardiectomy is the only definitive treatment option [11].
COVID 19 related acute complications like severe ARDS, acute kidney injury (AKI), and cardiac complications like acute Pericarditis are prevalent. Chronic complications like constrictive Pericarditis are also possible and leads to increased morbidity and mortality in COVID recovered patients.
