Author(s): Monica BUTNARIU
Diabetes is a heterogeneous syndrome, characterized by a complex disorder in the regulation of the body's energy metabolism, which affects the use of carbohydrates, lipids and proteins, as well as other metabolisms. These alterations result from an insulin-secreting defect associated with variable peripheral insulin resistance. The biochemical changes that these two disorders cause lead to functional cellular changes followed by irreversible anatomical lesions in many tissues and organs. Diabetes is the most common disease of the endocrine system and is triggered when the amount of insulin secreted in the body is not optimal or when peripheral cells do not respond to its action (insulin is a hormone that participates in lowering blood glucose). This condition causes disorders of the entire metabolism and, over time, can affect the functioning of various organs in the body
Diabetes mellitus (diabetes mellitus) is a metabolic disorder caused
by disruption of carbohydrate metabolism. Hyperglycemia, its
main feature, is determined by a secretory deficiency of insulin or
a resistance of the tissues to the action of this hormone. Statistical
data recently published by the World Health Organization (WHO)
reveal important aspects of the incidence and prevalence of the
disease, as well as its prognosis:
- In 2014, worldwide, 9 % of adults over 18 were diagnosed with
diabetes;
- Doubling the number of cases by 2030;
- Complications of diabetes (especially type II diabetes) are the
seventh leading cause of death worldwide; for example, in 2012,
1.5 million deaths worldwide occurred due to the micro- and
macrovascular complications of diabetes. Of these, about 50 %
were due to stroke, one of the most common complications of
diabetes;
- The prevalence of the disease varies depending on ethnic groups
and age, being more common in developed countries and after the
age of 60 years. In developing countries, the prevalence is lower
and predominates between the ages of 35-64 [1].
The whole world is facing a type 2 diabetes pandemic (diabetes mellitus), due to the westernization of the way of life, the aging population, urbanization, which result in changes in diet, adoption of a sedentary lifestyle and the development of obesity. The prevalence of DM differs significantly depending on the population studied, age, sex, socio-economic status and lifestyle. The predictions for 2025 are worrying and according to IDF / WHO estimates, the prevalence of DM will reach 9%. An important element, which in recent years has led to an increase in the incidence of the disease, has been the closer monitoring of the population and the improvement of diagnostic methods [2].
However, there are at least 30% of cases with undiagnosed type
2 diabetes. Type 2 diabetes accounts for 80-90% of all cases of
diabetes and is more common in overweight or obese people. He
often has a long asymptomatic period in which patients are not
diagnosed. There are studies showing that at the time of diagnosis,
more than half of the patients had one or more chronic complications
of diabetes. Diabetes is associated with a large number of chronic
complications whose end result is decreased quality of life and
premature mortality. Early diagnosis and treatment is the proposed
strategy for minimizing these effects. From an etiological point
of view, DM is a complex and multifactorial condition. Insulin
deficiency caused by the autoimmune destruction of Langerhans
Β cells and / or the body's resistance to the action of this hormone
are basic etiological mechanisms, unanimously recognized [3].
The biochemical defect in the secretion of incretins (intestinal
hormones secreted postprandially to stimulate insulin secretion),
genetic predisposition, increased stress-induced epinephrine
secretion, obesity-induced lipotoxicity and consequent insulin
deficiency are additional etiological mechanisms of diabetes.
Insulin is produced and secreted by the beta cells of the Langerhans Islands. Insulin is a polypeptide with a molecular weight of 5800 daltons, consisting of 2 amino acid chains: chain A (with 21 amino acids) and chain B (with 30 amino acids) linked together by 2 disulfide bridges (figure 1). The insulin molecule contains two polypeptide chains: A and B. Chain A consists of 21 amino acid residues, the first -glycol with the free -NH2 group, and the last-asparagine, having the free -COOH group [3].
The B chain consists of 30 amino acid residues, the first being phenylalanine, with the free -NH2 group, and the last is alanine with the free -COOH group.
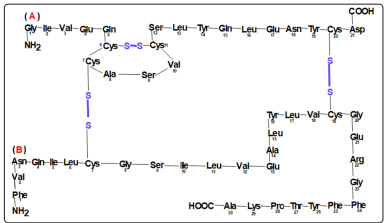
Figure 1: The amino acid sequence in the structure of insulin
In solution - depending on the concentration and pH is found in
the form of monomers, dimers and hexamers. The structure of the
chains differs depending on the species.
Human insulin differs from pig insulin only by the amino acid
at position 30 of the B chain: alanine in pigs and threonine in
humans. The cow is differentiated by 2 amino acids: in humans,
in position 8 of the A-threonine chain, in cow-alanine; in position
10 isoleucine in humans and valine in cows. Insulin biosynthesis
takes place in beta cell ribosomes.
It begins with the formation of preproinsulin - a 110 amino acid
polypeptide that is then cleaved into proinsulin (which has 86
amino acids). Proinsulin then undergoes a folding process in which
disulfide bridges are cleaved. Then the proinsulin under the action
of two endopeptidases is cleaved into insulin and peptide C. The
process of insulin biosynthesis ends with the formation of mature
secretory granules - which contain active insulin [4].
Then the actual secretion of insulin in the extracellular space
occurs by calcium-dependent exocytosis.
The most important factor that stimulates insulin secretion is
glucose. Once penetrated into the beta cell, it activates hexokinase,
which stimulates ATP production, which will close the K-ATPdependent channels.
Closure of K-ATP-dependent channels leads to depolarization
of the beta cell membrane and then to the opening of calcium
channels.
The physiological secretion of insulin is pulsating and oscillating.
The pulsations are of two types: some fast- which occur at intervals
of 6-10 minutes, with an amplitude of about 1.5 μU / mL; the
slower pulsations - at intervals of 90-120 minutes - give the
oscillating character of the secretion - consequent to the glycemic
variations.
On the other hand - insulin secretion caused by intravenous glucose
injection has two phases:
Phase I - with immediate onset, after 1-5 minutes, lasting about
10 minutes, during which insulin is released from the secretory
granules (also called and early phase) [5].
Phase II - with simultaneous onset but marked by phase Ia -
becoming evident after ten minutes - in which insulin is released
from both existing and newly synthesized secretory granules
lasting between 1 and 2 hours (also called late phase).
During physiological stimulation (a regular lunch) insulinemia
increases in the peripheral blood from 3-15μU / mL -basal
insulinemia- to 50-75 μU / mL. 60 % of the insulin secreted by
the pancreas is extracted from the liver in the first passage through
the portal vein to the other two insulin-dependent tissues: adipose
and muscle tissue.
Therefore - at the physiological stimulus, the insulin concentration
is 3-5 times higher in the portal vein than in the periphery.
The amount of insulin secreted by the pancreas in 24 hours is
about 48U. Half is basal insulinemia and the other half is prandial
insulinemia. Peptide C is secreted equimolarly with insulin,
resulting from the cleavage of proinsulin into insulin.
Because C-peptide is not captured by the liver, its peripheral levels
accurately reflect endogenous insulin secretion.
Under basal conditions, the concentration of peptide C is 0.4-2.2
ng / mL (on average 1ng / mL). After stimulation with glucagon
(1mg glucagon iv) peptide C increases approximately 20 times to
20ng / mL. In type 1 diabetes, basal C peptide is absent or below
0.4 ng / mL and does not increase after stimulation - demonstrating
absolute insulin deficiency.
Because C-peptide dosing does not interfere with insulin dosing,
it may be a good indicator of endogenous beta cell secretion in
patients with diabetes [6].
- hepatic gluconeogneza decreases;
- increases glycogenesis (in muscle, liver);
- decreases glycogenolysis;
- increases peripheral glucose utilization (90% in muscle and
10% in adipocyte);
- stimulates the active uptake of amino acids in muscle, liver
and fibroblasts (anabolic effect);
- antiproteolytic effect;
- increases hepatic lipogenesis;
- increases the esterification of fatty acids;
- activates lipoprotein lipase;
- diminishes the synthesis of ketone bodies in the liver;
- increases the use and hepatic clearance of ketone bodies [6].
The insulin preparations used in practice today are obtained by recombinant DNA technology and are chemically identical to human insulin. In addition to human insulin, insulin analogues (or "designer insulins") have been on the market for the past 15 years, which are human insulins with modified amino acid sequences to improve their pharmacological characteristics. Insulin monomers have a physiological tendency to aggregate and form "phenomenon polymers" that also exist in secretory granules. The majority ofhuman insulin preparations contain hexamers which after subcutaneous injection dissociate and are absorbed into monomers through the capillary wall [7].
From a pharmacokinetic point of view (ie in terms of absorption at the injection site), insulins are:
- Fast-acting insulins - lasting 6 hours;
- Insulin with very fast action and lasts 3-4 hours;
- Insulin with intermediate action and duration of 14-18 hours;
- Long-acting, 24-hour insulin;
- Biphasic insulins - mixtures of fast or very fast insulins with intermediate insulin with two peaks of action 2-3 hours and 6-10
hours after injection.
The faster absorption of insulin analogues (Humalog, Apidra, Novorapid) is given by changes in the amino acid sequence which
prevents their polymerization into stable solutions. As there is no longer the approximately 30-minute period required for dissociation
into monomers - their absorption begins at the time of injection - thus making their duration of action shorter, the insulin peak higher
than in the case of human insulin.
In the case of the first analogue recorded in 1996- Humalog (Lilly Company) reversed in position B28- B 29 proline with lysine, in
Novorapid (Novo Nordisk) it was replaced in position B28 proline with aspartic acid and in Apidra it was replaced in position B 29
lysine with glutamic acid. Table 1 shows some insulin preparations used and their time of action [8].
| No. | Type of insulin | Action starts | Maximum action | Duration of action |
|---|---|---|---|---|
| 1 | Very short-acting insulin: Humalog, Novorapid, Apidra | Immediate | 1 hour-1 and 1/2 hour | 3-4 hours |
| 2 | Short-acting insulin: Humulin R, Actrapid, Insuman R | 30 minutes | 2-3 hours | 6-8 hours |
| 3 | Intermediate insulin: Humulin N, Insulatard, Insuman basal | 2-4 hours | 6-10 hours | 14-18 hours |
| 4 | Long-acting insulin: Levemir, Lantus | 2-4 hours | No peak of action | 20-24 hours |
| 5 | Premixed insulins (Humamix 25, Huma mix 50, Mixtard 30, Novomix 30, Insuman Comb 25, Comb 50 etc.) | 10-20 minutes for those with human insulin analogs 0.5-1 hours for those with human insulin | 1-4 hours for those with analogues of insulin 2-10 hours those with human insulin | 20-24 hours |
Regarding the slow analogues, the changes to the biochemical
sequence of insulin are as follows: for insulin Lantus, asparagine
was replaced in position A21 with glycine and two arginine residues
were added at the C-terminus of the beta chain. The two arginine
residues change the precipitation point to the physiological pH and
the glycine in place of the aspergin stabilizes the hexamers. For
insulin Levemir, threonine was removed from position B30 and
myristic acid was attached to position B29-lysine. This determines
the binding to plasma albumin from which the product then
detaches at a relatively constant rate. Insulins are presented in
10 mL vials with 100u / mL or 3 mL cartridges with 100u / mL [9].
Cartridges are used to fill injection devices called "pens" - due
to their resemblance to a pen. There are also pre-filled pens.
Fast insulins and fast analogues are clear, transparent solutions.
Intermediate insulins are inhomogeneous-cloudy suspensions
with a tendency to precipitate; they must be resuspended before
injection. Biphasic or premixed insulins - being fast mixed
mixtures with intermediates are also inhomogeneous suspensions
- which require resuspension before injection [10].
Slow analogues are clear solutions that cannot be mixed with other
types of insulin. Insulin routes Insulin treatment is given with
subcutaneous insulin injections using special needles (0.5 cm, 0.8
cm, 1.2 cm long) attached to special insulin pens or syringes. The
most used regions are: abdominal wall, periombilical, deltoid region
of the arms, thighs, buttocks. Intramuscular administration may be
accidental - especially in children and people with poorly represented
adipose tissue if needles of inappropriate length are used [11-13].
The pharmacokinetics of insulin are thus altered - absorption is accelerated and the duration of action decreases. Intramuscular injection may be an alternative route to intravenous injection - but more painful and less safe. Insulin can be given intravenously for the treatment of ketoacidosis, hyperosmolar conditions and other critical conditions (acute myocardial infarction, stroke, parenteral nutrition) [14]. The administration is made by continuous infusion or bolus every 1-2 hours (given the short half-life of insulin). Side effects / side effects of insulin treatment:
1. Insulin allergy. Allergic reactions are very rare and can be
localized (erythema, paresthesias) or generalized (urticaria,
angioedema, anaphylactic shock); are mediated by Ig E
immunoglobulins and are attributed to the addition of
prothrombin or zinc, stabilizers or preservatives [15].
2. Injection lipodystrophy occurs when the injection technique
is defective and the injection is repeated at the same
site. It consists in the appearance of elastic nodules of
variable size. Repeated injections in the same area lead to
significant variability in absorption and pharmacodynamics.
Lipohypertrophy is reversible - if injection is avoided in that
area [16].
3. Refraction disorders. They are caused by glycemic fluctuations
at the beginning of insulin therapy and transient visual
disturbances [17].
4. Insulin edema is transient - more common in the lower limbs -
and very rarely generalized. It occurs due to the antinatriuretic
effect of insulin - especially in patients with a long period of
glycemic imbalance and significant dehydration [18].
5. Weight gain occurs at the beginning of insulin therapy and
is due to the disappearance of glycosuria. Significant weight
gain occurs as a result of nutritional therapy that does not meet
the patient's needs, the use of an inadequate insulin regimen,
and reduced exercise [19].
6. Hypoglycemia is the most common side effect of insulin
therapy. Decreased blood sugar below a certain level - difficult
to define in general - but around 50 mg / dL (<2.7-3.3 mmol
/ L) - causes the appearance of typical signs and symptoms
of adrenergic (sweating, palpitations, tremor, hunger) and /
or neuroglycopenic (headache, confusion, drowsiness, visual
disturbances, seizures) [20].
These signs and symptoms improve after glucose ingestion or
subcutaneous injection of glucagon or glucose infusion [21-23].
Hypoglycaemia also occurs with insulin therapy for the following
reasons:
- relative excess insulin (which inhibits hepatic glucose
production and stimulates peripheral glucose utilization in muscle
and adipose tissue);
- decrease or delay in food intake (which decreases the intake
of carbohydrates or precursors of neoglucogenesis);
- increased exercise (which accelerates the use of glucose in
muscle tissue);
- alcohol and drug use- by affecting hepatic gluconeogenesis
(alcohol) and by altering consciousness (alcohol, marijuana or
other illicit drugs) [24].
In terms of severity, hypoglycemia can be:
- Mild - with adrenergic-mediated symptoms (sweating,
palpitations, tremor, hunger), without cognitive deficit and
the patient is able to solve the episode by oral ingestion of
carbohydrates (10-15 grams) [25].
- Moderate - to which are added neuroglycopenic symptoms
(headache, behavioral disorders, confusion). The patient may need
assistance and treatment. Moderate reactions produce symptoms
that last longer and more severe - and often a second dose of
carbohydrates is needed [26].
- Severe- with loss of consciousness, convulsions or coma- the
patient needs emergency assistance with the administration of
subcutaneous or intramuscular glucagon or intravenous infusion
with hypertonic glucose [27].
Indications for Insulin Therapy:
- Type 1 diabetes. Insulin therapy in type 1 diabetes is a
substitution treatment- it is initiated at the time of diagnosis and
is continued for the rest of life. To this substitution treatment are
added nutritional therapy, self-monitoring, specific therapeutic
education and therapeutic physical exercise [28].
- Type 2 diabetes. Being a progressive disease- during its
evolution- insulin therapy will be added to the optimization of
lifestyle and oral medication [29]. Insulin therapy is also indicated
for type 2 diabetes in the following conditions:
- start with basal glycemia greater than 250 mg / dL and HbA1c<
9% with specific symptoms;
- peroperative;
- decompensation based on intercurrences (infections, traumas);
- acute myocardial infarction, acute stroke, heart failure. III-IV;
- pregnancy and lactation;
- contraindications to oral therapy (renal failure - clearance 50mL/
min or liver failure) [30].
Physiological secretion of insulin is characterized by the presence of a basal secretion (approximately 50% of total secretion) that inhibits hepatic glucose production and a prandial secretion following an increase in blood glucose due to ingestion. of food [31]. Ideally, exogenous insulin treatment should follow this pattern - which is clinically impossible with the preparations we have at hand. Therefore, this type of insulin therapy remains the closest compromise to physiological - to reach a normalization of blood sugar and maintain it [32].
Prandial insulin or bolus is best replaced by the injection of fastacting insulin or fast-acting analogue before meals (30 minutes for human insulin and before, during or after meals - for fast-acting analogue). It represents 40-60% of total daily doses [33].
The advantage of analogues over human insulin is obvious: the
possibility of injections during a meal or even after a meal - to
adjust the dose correctly according to the amount of carbohydrates,
shorter duration of action - which reduces the risk of hypoglycemia.
Noninferiority studies between human insulin and fast analogues
have not shown better metabolic control - with the use of fast
analogues - but the flexibility of the meal schedule and the amounts
of carbohydrates is an advantage.
Reproduction of basal insulin secretion (50%) can be done using
intermediate insulin in 1-2 doses or slow analogues-Lantus -1
dose and Levemir - usually in 2 doses. Using NPH in 1-2 doses
(before breakfast and before bedtime) involves over and / or
overnight hyperinsulinemia - which may require snacks to control
hypoglycaemia [34-36].
The use of slow analogues at bedtime reduces the risk of nocturnal
hypoglycaemia and in addition the individual variability of
absorption and therefore of pharmacodynamics is lower.
The most common basal bolus regimen is with a fast analog (or
fast human insulin) before meals - and a slow analog injection
(Lantus) in the evening at the same time.
Studies have shown that Lantus insulin can be injected at any time
of the day with the same results [37].
Its injection in the evening offers the possibility of its titration
using fasting blood glucose. Due to its shorter duration of action,
Levemir requires (in 45% of patients) two administrations per day
to approach the physiological secretion of basal insulin. Prandial
insulin is titrated according to the preprandial, postprandial
glycemias and the amount of carbohydrates in each meal [38-40].
The basal bolus regimen is indicated in the treatment of type 1
diabetes.
Studies have shown (DCCT - Diabetes Control and Complication
Trial, EDIC - Epidemiology of Diabetes Interventions and
Complications) that using basal insulin therapy bolus can
achieve and maintain near-normal glycemic targets with a
significant reduction (60%) in incidence and rate. progression
of microvascular complications (retinopathy, nephropathy and
diabetic neuropathy) [41].
The basal bolus regimen can also be used in type 2 diabetes
by progressively intensifying basal insulin therapy with the
same benefits and reducing the incidence and progression of
microvascular complications.
The risks of basal bolus treatment are hypoglycaemia - 3.5 times more common than during conventional therapy (with 2 injections of premixed insulin or 3 injections: 2 of fast-acting insulin andone of premixed insulin) [42-44].
However, the use of insulin analogues, the possibility of selfmonitoring, therapeutic education and access to specialized services reduce this risk.
The basal insulin therapy regimen is most commonly used to intensify the treatment of patients with type 2 diabetes on oral medication with metformin (or with oral medication - in double or triple combination of oral antidiabetics) and uncontrolled. The initiation of this insulin regimen is done with intermediate-acting insulin or analogue slowly at bedtime with 0.2 U / Kg body and titrated according to fasting blood glucose. The use of the analogue reduces the risk of nocturnal hypoglycaemia [45-47]. Glycosylated hemoglobin also decreases in this way until the therapeutic targets are reached.
This regimen aims to reduce postprandial glycemic excursions
after basal insulin has been titrated to fasting blood glucose in
therapeutic targets. That is, in a patient on basal insulin with
fasting blood glucose between 90 and 120 mg / dL we will monitor
postprandial blood glucose [48]. Depending on the lifestyle
(time and size of meals) to a glycosylated hemoglobin above the
therapeutic targets (which requires an intensification of treatment)
we can find postprandial blood glucose outside the therapeutic
targets, after breakfast or after lunch or after dinner. We will
correct this hyperglycemia with a quick analogue, injected before
the meal.
This type of treatment is relatively easily accepted by the patient
and is best initiated in the outpatient setting (in the particular
living conditions of the patient) [49-51].
He makes the transition to basal bolus treatment - if he will be
needed.
Premixed insulins are mixtures of fast-acting insulin or fast-acting
analogues with intermediate-acting insulins in varying proportions
(25/75, 30/70, 50/50). Premixed insulins are used in two injections
a day, every 12 hours, before breakfast and before dinner [52-54].
Their action being biphasic, fast-acting insulin (or fast-acting
analogue) covers the need for breakfast insulin, the interval
between breakfast and lunch, the peak of intermediate insulin
covers lunch and the rest - the interval until dinner; the pre-dinner
injection covers dinner and the night [55].
The disadvantage of this therapy is the difficulty of titration
(because both components will be titrated - fast and intermediate
simultaneously) and the need for meals with fixed amounts of
carbohydrates, at fixed hours, following the biphasic profile of
premixes. It also has an increased risk of hypoglycemia before
lunch (if the snack was missed or the meal was delayed) - less
often - but especially at night - in the first half of it [56].
Despite the disadvantages - it is an insulin regimen widely used
in type 2 diabetes - almost 40% and, unfortunately, in the case of
type 1 diabetes [57-65].
Elucidating the biochemical mechanisms of diabetes complications in order to prevent complications and develop an effective treatment for diabetes is an important step in preventing this disease. Despite the difference between the etiological mechanisms, the complications of the two types of diabetes (DZI and DZII) are the same and include microvascular, macrovascular and peripheral neuropathy complications. Although hyperglycemia is the determining factor in these complications, it influences microvascular complications more than macrovascular ones.
In the short term, hyperglycemia causes ketoacidosis and diabetic hyperosmotic syndrome or an increased predisposition to infections. In the long term, diabetes can develop with microvascular complications (retinopathy, nephropathy, peripheral and autonomic neuropathy) or macrovascular (deposition of atheroma plaques on large vessels at high risk of myocardial infarction, peripheral vascular disease, cerebrovascular disease). Gestational diabetes is associated with the new-born with an increased risk of complications or even infant mortality, and in the mother with obstetric complications and the risk of glucose intolerance.
Glucose intolerance is an entity associated with an increased risk of diabetes. About 20-50% of patients with glucose intolerance will develop some form of diabetes in the next 10 years, with one-third still returning to normal glucose tolerance. The risk of complications (especially cardiovascular) decreases considerably in the case of altered basal glycemic syndrome.
Diabetes is a disease that can significantly affect your lifestyle. To avoid complications, it is important that it is discovered in time, so it is ideal to perform regular tests to help detect any irregularities in the body. In addition, a healthy and balanced diet, complemented by regular involvement in sports, also support the proper functioning of the body.
